Massachusetts ALS Registry data
Data will be available on the Environmental Public Health Tracking website in late 2018. Visit the website for the ALS content page and future options to explore available data.
Reports and Data Briefs
- ALS Surveillance in Massachusetts: A one-of-a-kind registry for tracking an elusive disease
- ALS 2007-2011 Data Brief (DOC)
- Fact Sheet: The Prevalence of Amyotrophic Lateral Sclerosis and Multiple Sclerosis and Ecologic Evaluation of Selected Environmental Factors in Southeastern Massachusetts - October 2013 (DOC)
- ALS Feasibility Report (DOC)
- ALS in Massachusetts, 2008-2012
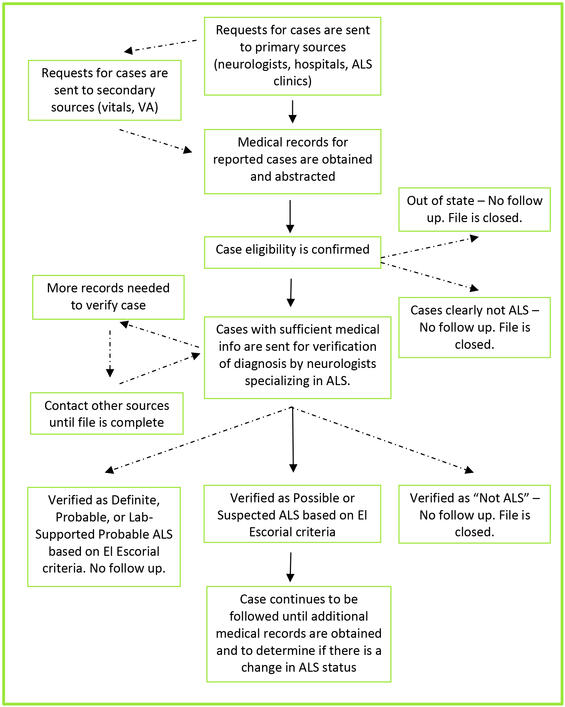
Case ascertainment overview
The MA ALS Registry aims to identify all eligible cases of ALS in Massachusetts on an annual basis. The eligibility criteria include residence in Massachusetts and a diagnosis of or treatment for ALS during a calendar year. State regulations require Massachusetts medical professionals to report ALS patients to DPH upon request. The privacy of all information collected for the Registry is protected by law. No patients are contacted by the Registry.
Patients do not self-register. All patients are reported by hospitals, clinics, and neurologists (primary sources). To be certain no patients are missed, patients are also identified from secondary sources that include ALS patient advocacy organizations, hospices, nursing homes, and death certificates.
Public health nurses, trained by consulting Registry neurologists, obtain and abstract records of the reported patients. The Registry receives an average of 600 reports annually on patients with a diagnosis of ALS. A one page abstract form is used to record all necessary clinical and demographic information from each medical record. These are first reviewed for eligibility. Clinical information is then reviewed for eligible patients and diagnoses are classified by a consulting neurologist under the revised, standardized El Escorial World Federation of Neurology Criteria for the Diagnosis of ALS.
Case ascertainment and verification flow chart
Additional Resources
-
Open PDF file, 111.53 KB, Case ascertainment and verification flow chart (English, PDF 111.53 KB)