Temperature
- Water temperature tells you how warm or cold the water is and is measured in degrees Fahrenheit (F) or Celsius (C).
- Water temperature is measured because it can influence many physical, biological, chemical and ecosystem processes in a waterbody. Temperature data helps to understand the temperature fluctuations and potential impacts to the overall water quality and ecosystem.
- Water temperature varies naturally throughout the day, season and depths for a waterbody.
Changes in water temperature
- Increased water temperature can result in decreased dissolved oxygen (DO) available for aquatic life, increased solubility of metals and other potential toxins in water, and presence of algal blooms.
- Both short- and long-term increases in temperature can negatively impact aquatic life in different ways. Short-term impacts are usually related to development and growth, reproduction and predation. Long-term impacts can be more significant with reduction or potential loss of aquatic species that are dependent on specific temperatures for survival and reproduction.
- Decreased water temperature can also result in impacts to aquatic life, such as decreased spawning in some fish and changes to egg hatching times and decreased metabolic rate of aquatic organisms.
What affects water temperature?
- Transfers of heat between a waterbody and the atmosphere, sunlight or incoming water source(s) affect the temperature.
- Water temperature is dependent on the weather, stormwater runoff, lack of vegetated shoreline shade, groundwater inflow, evaporation, and turbidity, among others.
- Waterbodies experience daily temperature changes.
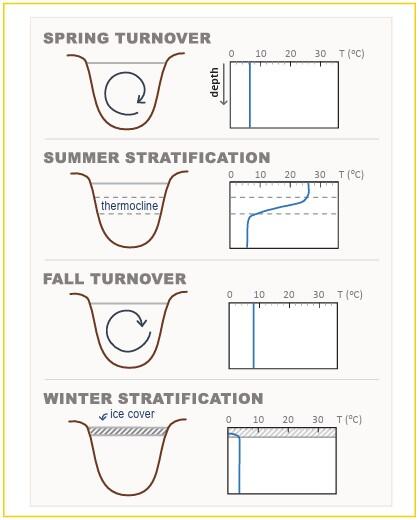
- Lakes and ponds also undergo seasonal temperature changes, by developing temperature layers or stratifying, in the summer and winter. During spring and fall, these temperature changes cause a waterbody to turnover and mix its entire water column to become a uniform temperature. (see figure)
Figure caption: Temperature layers and turnover in lakes by season. Adapted from Wetzel (1975). Courtesy of the US EPA (2021).
How is water temperature measured?
- Temperature is usually measured in the field with other water quality parameters such as pH, DO, and turbidity (using a Secchi disk).
- Temperature can be measured at varying intervals: either specific points in time or continuously. Specific points are usually measured with a handheld temperature probe (often in conjunction with other water quality parameters), whereas continuous monitoring is through floating or affixed temperature probes to understand the change of temperature through a day and/or over time to collect large data sets.
- If a water quality goal is to understand temperature trends in a waterbody, measurements should be taken at the same time of day. Measurements taken at different depths will provide information on vertical temperature changes, which can allow for understanding of stratification.
Dissolved Oxygen
- Dissolved oxygen (DO) is the amount of oxygen within the water that is available to aquatic organisms.
- DO is required for a waterbody to support aquatic life and is an important indicator of overall biological health of a waterbody.
- DO has a range of tolerance that various depending on the aquatic species, where some need higher amounts to survive while others can survive in lower DO conditions.
What affects dissolved oxygen?
- DO within a waterbody is primarily provided by transfer of oxygen from the air and by plants and algae.
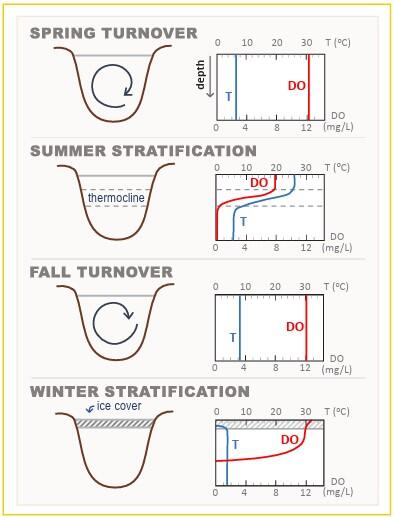
- Seasonal changes in water temperature affect DO concentrations that can be easily observed at the different stages of stratification. Colder water has higher concentrations of DO, whereas warmer water has lower concentrations.
- DO concentrations in surface water also vary by time of day. During the daytime, oxygen is added to the water by aquatic plants and algae photosynthesizing. At night, photosynthesis stops and DO levels decrease as oxygen is utilized by aquatic organisms.
- DO concentrations can also be associated with changes in other water quality parameters such as chlorophyll a, pH, and minerals.
How is dissolved oxygen measured?
- DO is usually measured in the field with other water quality parameters using a water quality probe and is reported in milligrams per liter (mg/L) and percent saturation.
- Low DO concentrations are considered hypoxic, while very low (virtually no oxygen) concentrations are considered anoxic.
- Similar to temperature, DO can be measured at specific points in time, or continuously to understand the DO changes through a day and/or over time.
- Also similar to temperature, DO can vary based on depth, so measurements should be at consistent times of day and at different depths to understand the vertical DO profile at a location.
pH
- pH is the concentration of hydrogen ions in water, and is measured to determine the acidity.
- pH values are on a scale from 0 to 14, with 7 being considered neutral. The pH scale is logarithmic, so everyone one-unit change in pH is a ten-fold change in acidity. pH plays an important role in chemical processes in waterbodies.
- Solutions with a pH below 7 are considered acidic, while those with a pH above 7 are considered basic. A change in pH can alter the concentrations and forms of chemicals in water, causing some otherwise non-toxic forms to become toxic for aquatic life.
- Different organisms have different pH levels in which they can survive, though they may need a specific pH level to develop or thrive.
What affects pH?
- pH in a waterbody fluctuates daily, with a higher pH during the day due to photosynthesis by aquatic plants and algae, and a lower pH at night when plants and algae are giving off CO2 instead.
- A well buffered waterbody, or a waterbody’s buffering capacity, is its ability to neutralize acids and bases and maintain a relatively stable pH.
- pH values are lower during seasonal times of rainfall and snowmelt because the excess incoming water does not get buffered through the ground soil.
- Local geology can help control pH, as some types of bedrock can buffer the water against acidic inputs.
How is pH measured?
- pH can be measured in the field using a water quality probe, indicator tests or strips, or from grab samples, but is usually measured via probe in the field with other water quality parameters.
- Similar to temperature and DO, pH can be measured at discrete times or continuously and should be measured at the same time each day if doing discrete measurements. Continuous monitoring allows for tracking pH changes throughout the day.
- pH can be variable because of it’s sensitivity to natural or human-induced factors, which can make it difficult to determine sources of trends or concerns.
Turbidity
- Turbidity is the measure of water clarity and is similar to total suspended solids (TSS). TSS is the concentration of suspended particles in water.
- An increase in turbidity can negatively affect aquatic life by clogging fish gills or filter-feeding systems, inhibiting visibility of prey for predators, decreasing light availability for aquatic plants, and altering life stage development.
- Increased turbidity is usually accompanied by higher water temperatures and reduced DO.
What affects turbidity?
- Turbidity can naturally increase by runoff from precipitation, bottom sediment disruption, small floating organisms suspended in the water column (cyanobacteria, plankton, etc.), and dead organic matter in the water column.
- Turbidity can also increase from human-induced factors like erosion, urban runoff, wastewater discharges, and bottom sediment disruption.
How is turbidity measured?
- Turbidity can be measured using a meter or sensor (reported in NTUs) or indirectly using a Secchi disk (reported in feet or meters).
- High turbidity levels don’t necessarily indicate poor water quality and low levels do not necessarily indicate good water quality.
Nutrients
- Nutrients support the growth of aquatic plants and algae which provide food and habitat for other aquatic life. Nitrogen and phosphorus are two of the most important nutrients in freshwater systems.
- Excess nutrients are one of the most common water quality issues affecting waterbodies. High nutrient concentrations are often from human activities and can negatively affect ecosystems. Most notably, excess nutrients can lead to excess biological growth, otherwise known as eutrophication, as well as cyanobacteria or harmful algae blooms (HABs).
What affects nutrients?
- Nutrients in waterbodies come from specific points (point sources) and from over general land areas (non-point sources). A point source could be a wastewater treatment facility that has discharges into a waterbody. A non-point source could be from fertilizer runoff or urban stormwater runoff.
- Nutrient concentrations can fluctuate throughout the year and usually have seasonal trends.
How are nutrients measured?
- Nutrients are measured by taking water samples in the field to be analyzed by a laboratory, and are commonly reported in milligrams per liter (mg/L) or micrograms per liter (ug/L).
- Sampling at different locations will provide different results depending on the suspected input source. Sampling should also be done throughout the year to understand seasonal variability.
- There are various forms of both nitrogen and phosphorus that can be analyzed for, though total nitrogen and total phosphorus provide insight into the overall amount of the nutrients.
E.coli and Similar Bacteria
- E.coli is a type of bacteria found in the intestines of people and animals, and thus found in their feces and can sometimes make people sick.
- E.coli (and similar bacteria) are measured from water samples to ensure water is safe for public recreation, as it is considered an indicator organism to identify fecal contamination in freshwater and indicate the presence of bacteria and viruses.
- Swimming in waters with elevated levels of E.coli causes an increased risk of getting sick from exposure to fecal viruses.
- More information on swim beach bacteria testing can be found on Massachusetts Department of Public Health’s Frequently Asked Questions About Monitoring Water Quality at Beaches website
What affects E.coli levels?
- E.coli from human feces can enter a waterbody from a wastewater treatment plant, broken or leaking sewer pipes, or failing septic systems.
- E.coli from animal feces can enter a waterbody from stormwater runoff anywhere there is wildlife.
How are E.coli measured?
- E.coli levels are measured by analyzing bacteria growth in a laboratory setting. Water samples are collected and brought to a lab for this analysis.
- Bacteria levels in waterbodies can be very variable, which makes determining a source of bacteria difficult.
More Water Quality Resources
Interactive Beach Water Quality Dashboard
Recreational Water Quality – Climate Hazard Adaptation Profile
DPH Pet Waste and Bathing Beaches
DCR Stormwater Design Handbook
DCR Stormwater Design Checklist
Stormwater Solutions for Homeowners: Rain Gardens
The Massachusetts Buffer Manual – Using Vegetated Buffers to Protect our Lakes and Rivers
Massachusetts Native Plant Palette
MassDEP Surface Water Quality Monitoring
SNEP Network Watershed Protection Standard Toolkit